"As part of our €56 million radiotherapy investment plan launched two years ago, the opening of the MRI-Linac to patients marks a major milestone," said Prof. Steven Le Gouill, Director of Institut Curie Hospital Group. " Today, Institut Curie—which boasts the most comprehensive radiotherapy technical platform in Europe—is reinforcing its leadership in the Paris region and beyond. The MRI-Linac is highly attractive to healthcare professionals and opens up new opportunities for research and clinical protocols on an international scale."
Combining Improved Quality of Life for Patients with More Precise Treatments
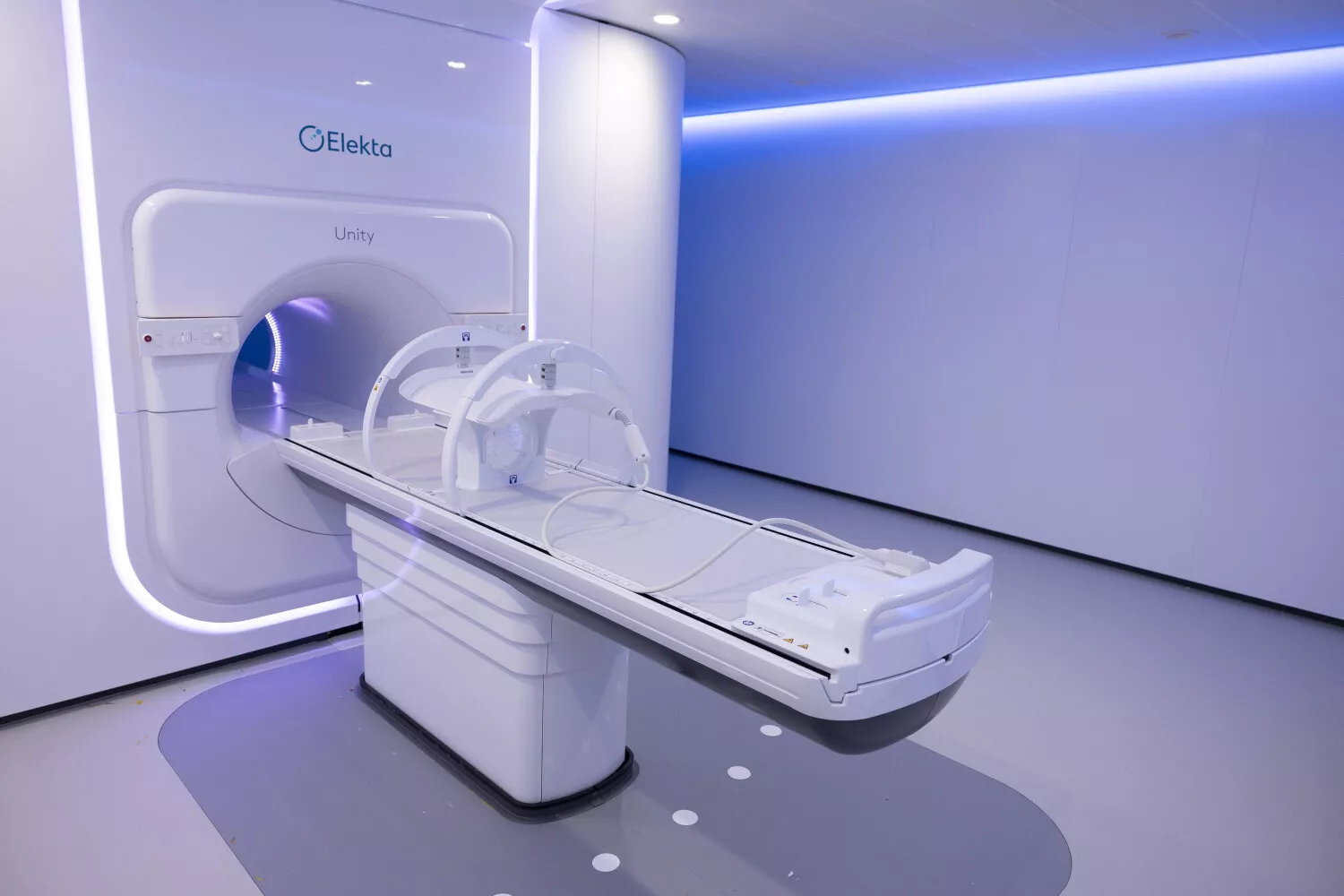
Elekta's MRI-Linac Unity combines a particle accelerator delivering radiotherapy with a high-field MRI (1.5 Tesla), enabling real-time monitoring of tumor evolution and internal organ movements. This unique capacity for instantaneous adaptation offers unmatched precision in treating complex cancers while preserving healthy tissue. Particularly effective for cancers such as prostate cancer, this technology accounts for the mobility of neighboring organs (such as the bladder or rectum) and adjusts radiation doses accordingly.
This precise, intensive targeting of the treatment area minimizes overall toxicity, preserves at-risk organs, and reduces the most undesirable side effects, significantly improving patients' quality of life.
Another major advantage is the significant reduction in the number of sessions required.
While classic radiotherapy may require up to eight weeks of daily treatment, MRI-Linac enables treatment completion in just five sessions over two weeks, with reduced toxicity.
"In the most complex, high-risk situations, MRI-Linac allows us to minimize toxicity risks by tracking tissue movements in real time during irradiation—a real benefit for patients' quality of life. This achievement results from a collaborative effort involving experts across various complementary medical specialties, each contributing to the lasting transformation of radiotherapy," said Prof. Gilles Créhange, Head of the department of oncological radiotherapy at Institut Curie.
A Collaborative Effort for the Benefit of Patients
The installation of the MRI-Linac at the Saint-Cloud hospital site represents the culmination of a long-term project demonstrating Institut Curie's commitment to uniting multidisciplinary teams around cutting-edge innovations. Biomedical engineers, medical physicists, radiologic technologists, clinicians, and dosimetrists worked together to bring this ambitious project to life.
 |
 |
|
Key figures and info: > More than 245,000 patients treated by radiotherapy each year in France (French National Cancer Institute (INCa) - Panorama des cancers 2024) |